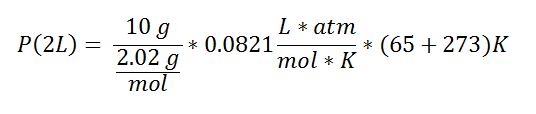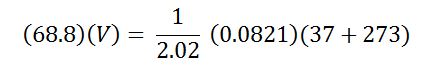Applications of the Ideal Gas Laws
The ideal gas laws are best applicable at high temperatures and low pressures. At these conditions, gases move the most effectively across their construct and most accurately follows the principles of the kinetic molecular theory. By combining the gas laws of Boyle, Charles, Gay-Lussac, and Avogadro,
Example
10 g of H2 gas is placed in a 2.00L pressurized container at a temperature of 65°C. There was a leak that was patched up a few minutes later. There was only 1 gram that remained and the temperature was decreased to 37°C. Assuming the pressure remained the same, what is the final volume of the container?
Solution:
Use the Ideal Gas Law PV = nRT
Solution:
Use the Ideal Gas Law PV = nRT
After solving this equation, we obtain P = 68.688 atmospheres. We must then use PV = nRT a second time in order to represent the final state of the container. We can now substitute the value of P into the equation.
The final volume thus equals 0.183L or 183mL
Practice Problems
1. 32 grams of Sulfur Dioxide is added with 32 grams of Oxygen gas to produce Sulfur Trioxide. What is the Volume if the temperature of the flask is at STP?
2. 745 g of gaseous uranium hexafluoride (UF6) is compressed into the One Ring, which has a volume of 0.75L. The band is then thrown into the fires of Mt. Doom, where the temperature reaches 9001°F. If the Ring can withstand pressures of up to 1000 atmospheres, will Sauron be defeated?
3. Grace Zhang makes instant ramen in a bowl and closes the lid. Of course, she is caught by Schnell, and is made to clean the lab tables. If the pressure of the gas over the ramen decreases from 1.6 atm to 1.2 atm, what is the change in temperature of the gas (assume the partial pressure of the ramen is 0)?
4. Singed keeps a bag of noxious gases on his back. The pressure inside this bag is 2.3 atmospheres, and it can hold a maximum of 1337 L of gas. If Singed raises the temperature of the gas to 564 K, and releases the gas at rate of 10 mol/s, how many seconds can Singed continue releasing gas?
Solutions
2. 745 g of gaseous uranium hexafluoride (UF6) is compressed into the One Ring, which has a volume of 0.75L. The band is then thrown into the fires of Mt. Doom, where the temperature reaches 9001°F. If the Ring can withstand pressures of up to 1000 atmospheres, will Sauron be defeated?
3. Grace Zhang makes instant ramen in a bowl and closes the lid. Of course, she is caught by Schnell, and is made to clean the lab tables. If the pressure of the gas over the ramen decreases from 1.6 atm to 1.2 atm, what is the change in temperature of the gas (assume the partial pressure of the ramen is 0)?
4. Singed keeps a bag of noxious gases on his back. The pressure inside this bag is 2.3 atmospheres, and it can hold a maximum of 1337 L of gas. If Singed raises the temperature of the gas to 564 K, and releases the gas at rate of 10 mol/s, how many seconds can Singed continue releasing gas?
Solutions