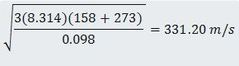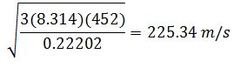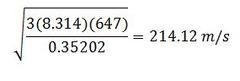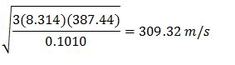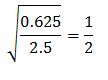KMT Solutions
Root Mean Square Velocity
1. Find the root mean square velocity of molecules of gaseous phosphoric acid at 158°C (its boiling point).
Use the formula for urms. M = the molar mass of H3PO4 in kilograms = 0.098 kg.
Use the formula for urms. M = the molar mass of H3PO4 in kilograms = 0.098 kg.
2.
Find the root mean square velocity of radon at 452K
M for radon is .22202
M for radon is .22202
3.
Find the root mean square velocity of uranium hexafluoride at 647 K.
M for uranium hexafluoride is .35202 kg
M for uranium hexafluoride is .35202 kg
4.
Find the root mean square velocity of dichlorosilane (H2Cl2Si) at 238°F
M for dichlorosilane is .1010
T = (5 / 9) (238°F - 32) + 273 = 387.44 K
M for dichlorosilane is .1010
T = (5 / 9) (238°F - 32) + 273 = 387.44 K
Effusion and Diffusion
1. 20.0g Oxygen gas = 0.625 moles
10.0g Hydrogen gas = 2.5 moles
10.0g Hydrogen gas = 2.5 moles
1 mole Oxygen gas : 2 mole Hydrogen gas
2.
2.
3. It will be a tie
4. Element Y will win.
4. Element Y will win.