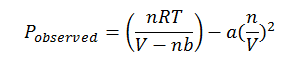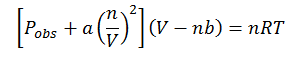The Real Gas Law
The Ideal Gas Law is a quick and easy way to calculate the approximate values of volume, pressure, temperature, or number of moles in a sample of gas, but at higher pressures, or when greater accuracy is desired, the real gas law must be employed.
The real gas law utilizes the same framework as the Ideal Gas Law, but with two important correction factors included for volume and pressure
The real gas law utilizes the same framework as the Ideal Gas Law, but with two important correction factors included for volume and pressure
At higher pressures, PV / RT begins deviating greatly from ideal conditions
One reason the Ideal Gas Law does not fully model the behavior of gases is because it does not take into account the volume occupied by the gas molecules themselves.
To rectify this error, the formula for volume in the Real Gas Law is:
Vcorrected = Vcontainer - nb
where b is a characteristic factor and n is the number of moles of gas.
To rectify this error, the formula for volume in the Real Gas Law is:
Vcorrected = Vcontainer - nb
where b is a characteristic factor and n is the number of moles of gas.
The other flaw in the Ideal Gas Law is its failure to account for the interparticle attractions betweens gas molecules. Because of these interparticle attractions, the actual pressure exerted by the gas hitting the walls of the container is less than that calculated by the ideal gas law. Because the amount of pressure reduced is proportionate to the concentration of pairs of interacting gas molecules per unit volume, the corrected formula for pressure is:
Thus, the overall real gas law can be written as:
This is called the van der Waals equation.
Link to some of the van der waals constants (a & b): http://www2.ucdsb.on.ca/tiss/stretton/database/van_der_waals_constants.html
Practice Problems
1. Find the final pressure if there is 10 grams of Oxygen gas with 0.5L at 50°C.
2.Find the final temperature if there are 14 grams of Nitrogen gas in a 500mL beaker at 600torr.
3. Find the temperature of gas if there is a 2 moles of Carbon Dioxide in a 1L flask with 1 atm at 0°C.
4. A quantity of aluminum trichloride (AlCl3) is added to a 4 L rigid container at 567 K and 23 atm. Calculate the moles of gas using the Real Gas Law.
2.Find the final temperature if there are 14 grams of Nitrogen gas in a 500mL beaker at 600torr.
3. Find the temperature of gas if there is a 2 moles of Carbon Dioxide in a 1L flask with 1 atm at 0°C.
4. A quantity of aluminum trichloride (AlCl3) is added to a 4 L rigid container at 567 K and 23 atm. Calculate the moles of gas using the Real Gas Law.