Pressure-Temperature Relationship in Gases Lab

Full Lab Procedures and Materials are accessible here:
http://www2.vernier.com/sample_labs/CWV-07-COMP-pressure_temperature.pdf
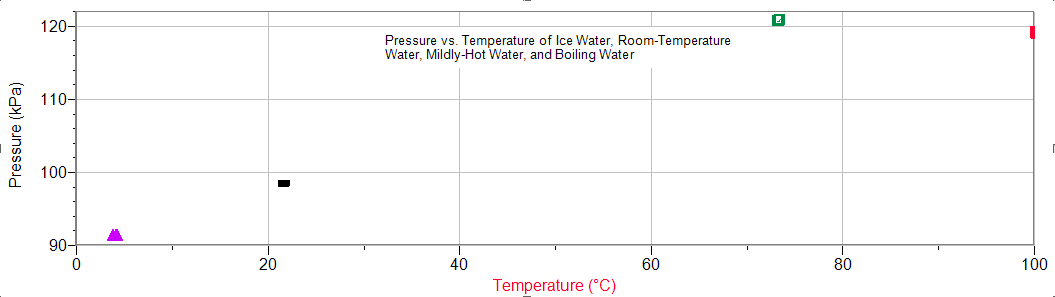
The above data shows a positive and relatively linear association between pressure and temperature. It is important to notice that the pressure at 73 degrees Celcius is higher than the pressure at 100 degrees Celcius. This can be explained by the fully submerged beaker during our measurement of the pressure of mildly-hot water and the only half-submerged beaker during our measurement of the pressure of boiling water. To boil water, a hot plate was heating the beaker and its contents at its maximum possible temperature, making submerging the flask with our bare hands dangerous and requiring us to use a ring clamp to suspend the flask into the beaker. Because a 1500 mL beaker was used, it was impossible to lower the entire flask into the beaker and thus creating an amalgram of room temperature air and boiling hot water surrounding the beaker. Although the temperature of the boiling hot water read 100 degrees Celcius, the flask did not, because of the surrounding room temperature air, thus creating a lower reading than the mildly-hot water.
To see the full results of the lab, click the link below:
To see the full results of the lab, click the link below:
| pressure_vs._temperature_lab.pdf | |
| File Size: | 371 kb |
| File Type: | |