Avogadro's Law
What is Avogadro's Law?
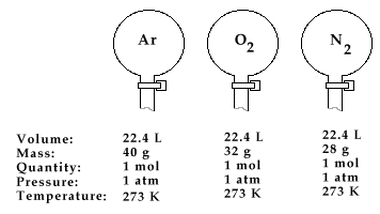
Avogadro's Law states that at the same temperature and pressure, the volume of an ideal gas are directly proportional to the number of moles, n. This can be represented mathematically by the equation P = kn. A direct consequence of Avogadro's Law is that 1 mole of any ideal gas occupies a volume of 22.4 L at STP. At higher pressures, the calculated volume begins to deviate more from the experimental result.
Example
A beaker is filled with 8 grams of Oxygen gas in a fixed volume of 2 Liters. One of the valves opened and there remains only 2 grams of Oxygen gas, what is the new volume of gas?
8 grams Oxygen gas = 0.5 moles Oxygen gas
2 grams Oxygen gas = 0.125 moles Oxygen gas
V1/n1 =V2/n2
2 Liters/0.5 moles = x Liters/0.125 moles
x = 0.5 Liters = 500 mL
8 grams Oxygen gas = 0.5 moles Oxygen gas
2 grams Oxygen gas = 0.125 moles Oxygen gas
V1/n1 =V2/n2
2 Liters/0.5 moles = x Liters/0.125 moles
x = 0.5 Liters = 500 mL
Sample Problems
1.
What volume does 35.6 grams of xenon tetrafluoride occupy at STP?
2. A 250mL rigid flask contains 40 grams of carbon dioxide. A plug was placed to close the opening, but as Bob tried to close it, he was temporarily distracted and let some amount of carbon dioxide go. He remeasured the Volume and found it to be 90 mL. How much, in kg, of the carbon dioxide was released?
3. Assume that it requires the ratio of 100 moles of He to a 213L tunnel to lift a human being (probably not true and assume pressure is constant along with no other factors affecting the tunnel are present). The tunnel is modified to be smaller and was shaped into a 115L tunnel and 80 moles of He was present. Does the modified tunnel have the ability to lift a human being?
4. A balloon has an initial volume of 150.0mL of gas and 4.00 grams of helium along with 17.73 grams of Chlorine gas. Some gas dissipated and some added totaling the moles of combined gas is 1 mole. What is the final volume? If there is 1 gram of Helium, how many grams of Chlorine gas exists after?
2. A 250mL rigid flask contains 40 grams of carbon dioxide. A plug was placed to close the opening, but as Bob tried to close it, he was temporarily distracted and let some amount of carbon dioxide go. He remeasured the Volume and found it to be 90 mL. How much, in kg, of the carbon dioxide was released?
3. Assume that it requires the ratio of 100 moles of He to a 213L tunnel to lift a human being (probably not true and assume pressure is constant along with no other factors affecting the tunnel are present). The tunnel is modified to be smaller and was shaped into a 115L tunnel and 80 moles of He was present. Does the modified tunnel have the ability to lift a human being?
4. A balloon has an initial volume of 150.0mL of gas and 4.00 grams of helium along with 17.73 grams of Chlorine gas. Some gas dissipated and some added totaling the moles of combined gas is 1 mole. What is the final volume? If there is 1 gram of Helium, how many grams of Chlorine gas exists after?